TRACER
Clinical Trial Feasibility Analysis Tool
Synthesize multi-registry data in minutes. From trial registries to actionable insights using AI-Assisted clinical trial landscape analysis.
What is TRACER?
Smarter Protocol Design and Feasibility Planning
TRACER is an AI-assisted, registry-centric platform that helps sponsors and research teams rapidly understand the competitive trial landscape for any therapeutic area.
Why Feasibility Analysis Matters?
- Clinical trials face high failure rates due to recruitment challenges and protocol assumptions that don't hold up in execution.
- Traditional landscape reviews are manual, time-consuming, and often limited to a single registry or bibliographic databases.
Advantages of TRACER
- Provides a faster, more comprehensive starting point.
- Automatically synthesize data across multiple trial registries and augment with linked publication records.
Benefits of TRACER
- Protocol teams can surface blind spots earlier and enter feasibility discussions with evidence, not assumptions.
- Supports smarter concept development, protocol design, and faster go/no-go decisions.
What Can You Do With TRACER?
Whether you’re evaluating a new indication, benchmarking your protocol design, or preparing for an advisory board, TRACER synthesizes trial registry and publication data into structured, defensible summaries
Competitive Landscape
See who else is running trials in your space, what phases they're in, and where they're enrolling, across all primary registries.
Design Benchmarks
Compare your planned sample size, endpoints, and eligibility criteria against what similar trials have used.
Reduce Analysis Time
Manual searches typically cover one registry at a time. TRACER automates multi-registry retrieval, deduplication, and synthesis, delivering structured summaries with publication context in a fraction of the time.
Features of TRACER
- Synthesize data from multiple registries.
- Includes ClinicalTrials.gov and primary registries indexed by WHO ICTRP.
- Augmenting with PubMed publication data.
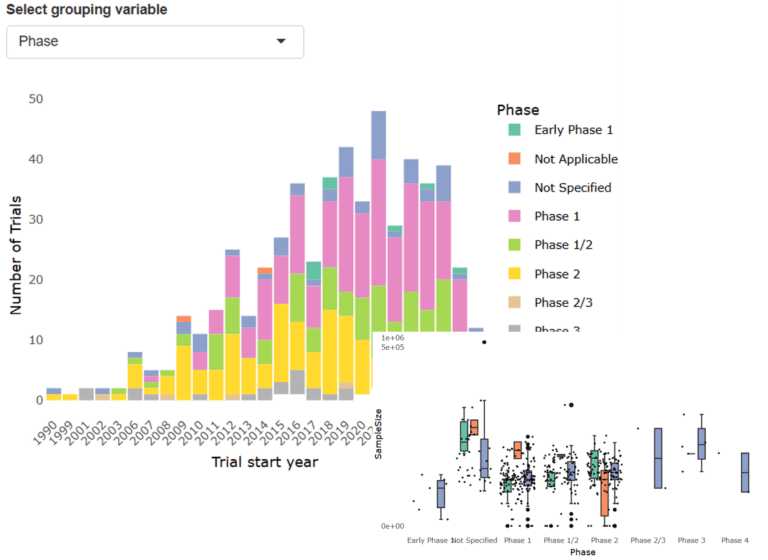
- Interactive Visualizations: Explore trends and patterns through automated charts and dashboards.
- Timeline Insights: Benchmark expected enrollment durations against completed trials.
- Identify design patterns, enrollment benchmarks, and endpoint conventions.
- Export sponsor-ready summaries and briefing documents.
TRACER complements expert judgment;
it doesn't replace it.
Provides A Faster, More Comprehensive Starting Point
TRACER is a decision-support tool that accelerates the early stages of landscape review. It helps you explore the competitive landscape quickly.
Protocol Planning Requires Expert Involvement
A submission-ready protocol requires expert input, accounting for regulatory requirements, population heterogeneity, and operational realities.
Reduce Feasibility Risk By Leveraging Expert Analysis
Don't build your protocol on untested assumptions. Discuss with our experienced biostatistics and clinical operations team. We can help you interpret landscape data and translate it into a defensible study design.
When you’re ready to move from landscape review to protocol planning, contact EvoClinical to discuss your next study.
Clinical Trial Feasibility Analysis
FAQs
What is clinical trial landscape analysis?
Clinical trial landscape analysis is the process of systematically reviewing ongoing and completed trials in a therapeutic area to understand competitive positioning, common design patterns, and enrollment benchmarks.
What are the benefits of Clinical Trial Landscape Analysis?
Understanding what similar trials have done (and how they performed) helps sponsors avoid common pitfalls, set realistic enrollment expectations, and differentiate their study design.
What clinical trial data sources does TRACER use?
TRACER synthesizes clinical trial data from multiple public trial registries (including ClinicalTrials.gov and primary registries indexed by WHO ICTRP) and augments with linked PubMed publication data for comprehensive coverage.
How is TRACER different from manual registry searches?
Manual searches typically cover one registry at a time. TRACER automates multi-registry retrieval, deduplication, and synthesis, delivering structured summaries with publication context in a fraction of the time.
Does TRACER provide data visualizations?
Yes. TRACER includes automated charts and dashboards that visualize trial landscape trends, making it easier to interpret patterns and communicate findings to stakeholders.
Is TRACER a replacement for clinical trial feasibility consultants?
No. TRACER is a decision-support tool that accelerates the early stages of landscape review. For full feasibility assessments, we recommend engaging with our expert team.
What outputs does TRACER provide?
TRACER generates interactive visualizations, structured summaries, benchmark reports, and sponsor-ready export documents suitable for internal decision-making and advisory discussions.
How current is the trial data in TRACER?
TRACER pulls from continuously updated public registries and PubMed, ensuring access to the most recent available trial records and publications.
Does EvoClinical provide Clinical Trial Feasibility Analysis Consulting?
Yes. At EvoClinical, we go beyond traditional feasibility by leveraging advanced tools that combine public trial registry data, landscape analysis, and predictive modeling. Our platform delivers rapid, data-driven insights, enrollment feasibility, and investigator networks, empowering sponsors to make smarter, faster decisions in the bid-development cycle.